 
The third structure has a -2 charge on the nitrogen atom and the second structure has a -1 on the nitrogen atom which is less electronegative than oxygen. The formal charges add to -1 and the more negative formal charge is on the more electronegative element. The first structure is the most reasonable structure. If it is an ion, the formal charges must add to the charge on the ion. If it is a neutral compound, formal charges must add to zero. The formal charges must add to the charge on the species. We need to determine which is the most reasonable Lewis structure fo the OCN – ion.įirst, determine the formal charges on all of the atoms in the three structures. A more positive formal charge should be on the less electronegative atom. A more negative formal charge should be on the more electronegative atom.Ĥ. The same nonzero formal charges on adjacent atoms are not preferred.ģ. 
For example a -1 is preferable to a -2 and a +1 is preferable to a +2.Ģ. Smaller formal charges (absolute values) are preferable to larger ones. Here are the rules for using formal charge to choose the best Lewis structure.ġ. There are times when one needs to determine a more important resonance structure or determine how the atoms are connected in some compounds. The nitrogen has a formal charge of +1, and the hydrogen atoms have a formal charge equal to zero. The neutral nitrogen atom has 5 valence electrons, and the atom in the compound owns 4 electrons. First draw the Lewis structure, and then count the number of electrons owned by nitrogen in the compound. Circle the electrons around hydrogen, and we see each of the hydrogens own one electron. The formal charges are written next to the atom and circled.Īnother way to do this is to draw the Lewis structure and replace the single bonds with the bonding electrons.Ĭircle the electrons around the carbon. The hydrogens each own 1 electron, andīoth carbon and each of the 4 hydrogens in methane have a formal charge of zero. 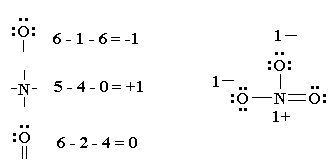
The number of valence electrons for the neutral carbon atom is 4. Then count the number of bonding electrons. Then count the number of lone pairs on the carbon atom. Let’s look at the formal charges for the atoms in methane. # valence e – in neutral atom – the # e – owned by the atom in the moleculeĪn atom in a molecule owns half of its bonding electrons and it owns its lone pairs (or even a single electron as in a free radical). Formal charges provide a way for one to determine the most reasonable or best Lewis structure as well as the more likely structure if we are not sure how the atoms are connected. The formal charge is the number of valence electrons in a neutral atom minus the number of electrons the atom owns in the compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
